What type of metal were the plates made of?
While the text of the Book of Mormon itself does not specify the composition of the plates, except that they were made of "ore", nineteenth-century witnesses described them as having the “appearance of gold.” William Smith was the only witness to say that the plates were made of a gold and copper alloy, although it is unclear how or why he came to this conclusion. Examples of gold and copper alloys capable of being worked to resemble pure gold have been discovered in both the Old and New Worlds. These findings provide historical context for the possibility that such an alloy was used in the creation of the plates.
Artifacts of gold and copper alloy have been found in the New World dating as far back as the second millennium and in the Old World dating to the third millennium BCE. Artifacts made from this alloy were typically treated to make the surface of the object have the "appearance of gold" through various methods of depletion gilding.
Depletion gilding is the process of exposing the surface to some form of corrosive material that attacks the surface level of copper and leaves the gold, which is then burnished and made to have the appearance of pure gold. Gold and copper alloys appear similar in color to copper, but after depletion gilding, the surface appears similar to pure gold.
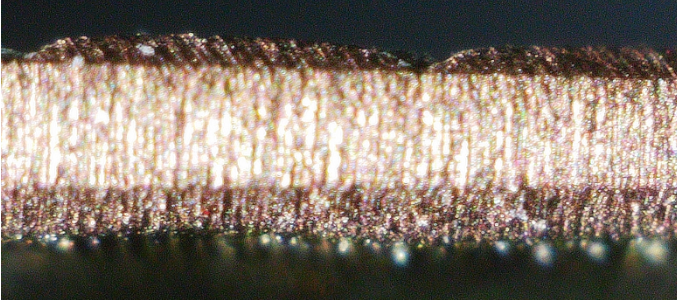


The Spaniards who colonized the New World referred to Mesoamerican gold-copper or gold-copper-silver alloys as tumbaga. The process of depletion gilding of tumbaga was noted by Gonzalo Fernandez de Oviedo (1535–1548), who wrote:
"...the indigenous people of Mexico 'know very well how to gild the objects and items they make from copper and low grade gold. And they have such ability and excellence in this, and give such a high lustre to what they gild, that it looks like good gold of 23 carats or more. . . . They do this with a certain herb, and it is such a secret that any goldsmith in Europe or in any other part of Christendom, would soon become a rich man from this manner of gilding.'"
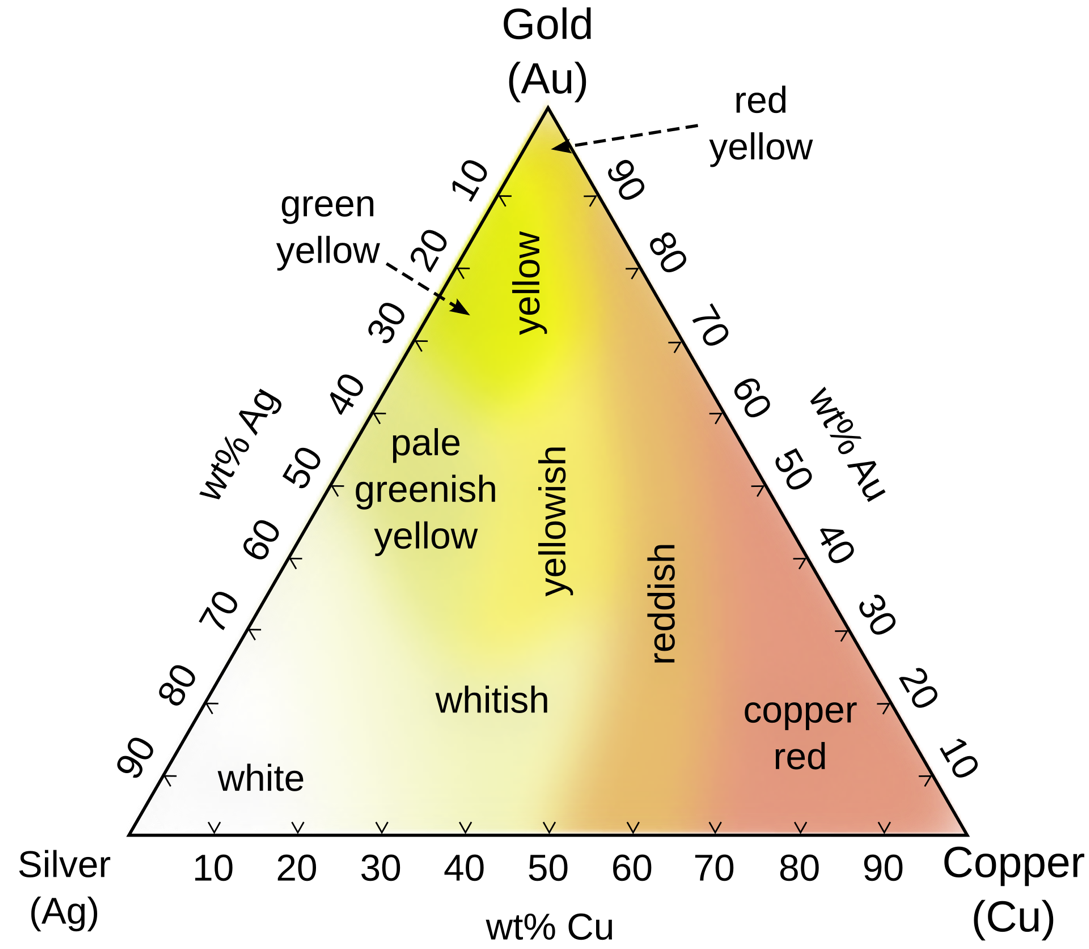
After the depletion gilding process, the color of the surface of the object is determined by the remaining elements, which are primarily gold and silver, with some copper remaining in tumbaga if it is copper-rich. Depending on the concentrations of gold, silver, and copper that the depletion gilding leaves on the surface, the gold color of the tumbaga may be whitish, yellowish, reddish, or even greenish in appearance.
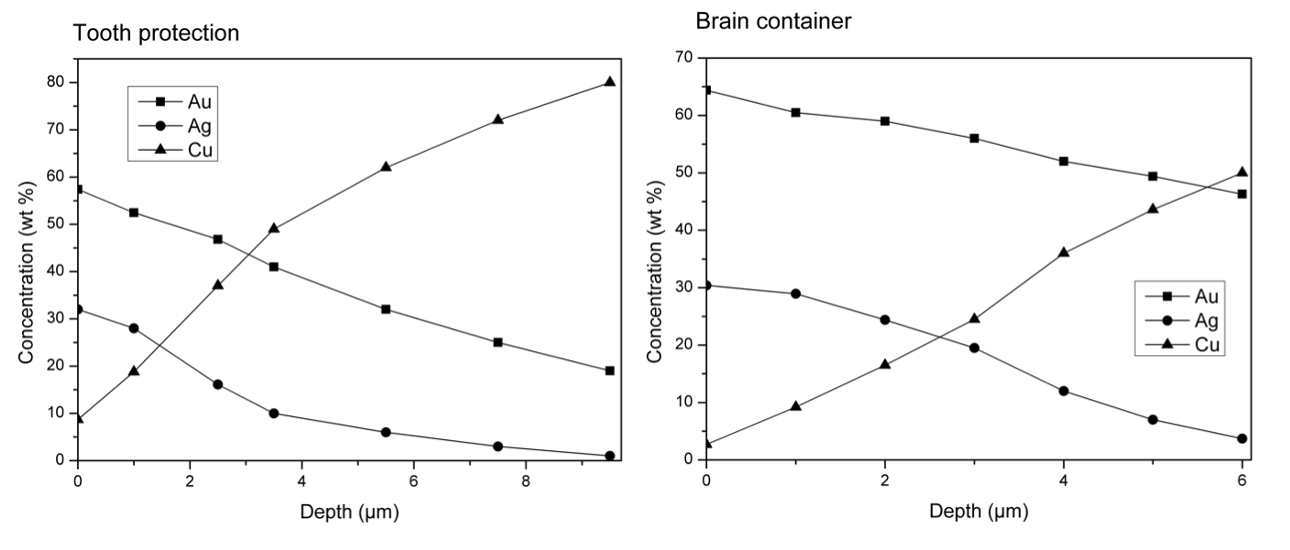
In considering the elemental makeup of the plates, the calculation includes the copper, gold, and silver, but the ratio of silver is fixed to be 10% of the gold. For example, if a valid combination of the physical properties of the plates has Gold Alloy Content at 65%, then the silver content will be set to 6.5%. The rationale for this constraint is twofold. The first consideration is that ancient depletion gilding was only able to remove copper and not silver, and when high concentrations of silver are present in a tumbaga alloy, the color no longer has "the appearance of gold" but tends to appear greenish or white. The second consideration is that while there are many examples of ternary alloys of tumbaga artifacts of varying concentrations of gold, silver, and copper, many tumbaga artifacts are essentially binary alloys with just trace amounts of silver.
Silver is marginally more dense than copper (per cubic inch, silver is 171.9 grams, and copper is 146.5 grams, whereas gold is 316.6 grams), so minimizing the amount of silver in calculations will not have a significant impact on the overall results compared with variations in gold content. However, more precision may be gained in calculations that include a greater variation in silver content.